
Correlation between drug-induced sleep endoscopy criteria for patient selection and transoral robotic surgery outcomes in obstructive sleep apnea treatment
Introduction
Obstructive sleep apnea syndrome (OSAS) is one of the most common type of respiratory sleep disorder, with a prevalence of up to 30% in adults (1,2). This is a progressive disease, as well as a public health burden, which increases the risk of traffic and occupational accidents, triggering the development of metabolic syndrome, having neurocognitive and cardiovascular consequences, such as acute myocardial infarction, stroke and hypertension (3,4).
Continuous positive airway pressure (CPAP) is the gold standard treatment, whose efficacy has been scientifically proven. Nevertheless, its compliance and adherence rates being under 50% in long-term follow-up (5) justifies the current quest for other treatment options.
The most performed surgical procedure to treat OSA is uvulopalatopharyngoplasty (UPPP), but systematic reviews have demonstrated a success rate of only 45%, highlighting the need for improvement in the surgical indication for OSA treatment (6).
Transoral robotic surgery (TORS) was described to treat the tongue base obstruction, appointed as responsible for the failures of the pharyngeal surgeries. The tongue base reduction (TBR) has demonstrated promising results, when combined with the expansion sphincter pharyngoplasty (ESP), treating simultaneously the lateral pharyngeal wall and the tongue base obstruction (7). However, the current literature still presents variable results, which can be attributed to the criteria adopted to select the candidates for surgical treatment in each study (8).
Patient selection based exclusively on physical examination characteristics (9,10), such as tonsils size, palatal position (modified Mallampati) and body mass index may explain such variability regarding the surgical treatment outcomes, once the information obtained under awake conditions underestimate the neuromuscular relaxation occurred during sleep and consequently its impact in OSA physiopathology (11).
Drug-induced sleep endoscopy (DISE) is a complementary test that is able to reproduce the muscular relaxation occurred during sleep (12), allowing a dynamic evaluation of the upper airway collapse that causes apnea (13) and the detection of obstructive sites that diverges from those observed during awake evaluation (14,15). However, its value in selecting patients for surgical treatment is not well established and studies are still needed to demonstrate if DISE can improve surgical outcomes.
Assuming the theory that an impaired genioglossus activity is a paramount factor in OSA physiopathology, the lack of its muscular support to the tongue will causes retroglossal obstruction (16). In our hypothesis, DISE can improve TORS outcomes in OSA treatment if used to detect the tongue base obstruction due to the lack of muscular support, contraindicating the surgical procedure in these patients.
Objective
The objective of this study is to correlate the DISE criteria adopted for patient selection to TORS in OSA treatment with the surgical outcomes.
Methods
This is a retrospective case series analysis of adult individuals submitted to DISE as candidates to OSA surgical treatment, from January 2016 to December 2016. The research ethics committee approved the trial under number 52479116.3.0000.5487.
OSA was diagnosed with type 1 polysomnography (PSG), scored by a sleep medicine doctor blind to the surgical treatment, according to the 2012 American Academy of Sleep Medicine (AASM) scoring manual.
The surgical treatment was considered in patients with apnea-hypopnea index (AHI) >15 events/hour and non-compliant to CPAP. Physical examination was performed in all patients in order to evaluate the modified Mallampati, the palatine tonsils size, the lingual tonsils size and BMI (10). Patients with BMI >40, cranio-facial deformities, palatine tonsil hypertrophy (3 and 4), age over 60 years or with any clinical contra-indication for a surgical procedure were excluded.
DISE was performed before the surgery in all candidates for surgical treatment, using Propofol delivered through a target-controlled infusion pump (TCI®) in the presence of an anesthesiologist, following the protocol described by Rabelo et al., which is able to reproduce the AHI from control PSG (17). The tests were performed in an endoscopy center by a trained ENT doctor and the images were recorded and digitized for subsequent evaluation and classification according to the VOTE scoring system (13).
The test was proposed to improve detection of patients with retroglossal obstruction due to the lack of muscular support, excluding patients presenting this type of obstruction. Therefore, it was primarily used to contraindicate the procedure in the group in which the obstruction mainly occurred because of the muscle relaxation.
The way to help in identifying this type of obstruction is to observe the improvement of retroglossal obstruction after gently closing the mouth, without any mandibular advancement or neck hyperextension, with the objective of making the tongue assume its proper position under the hard palate (Figures 1 and 2). After performing this maneuver, patients with persistent retroglossal obstruction were considered as having soft tissue hypertrophy and were selected to undergo surgery (Figure 3).
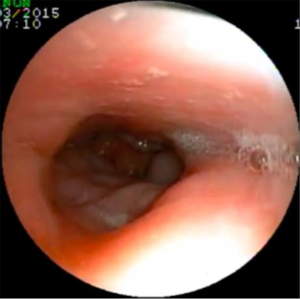

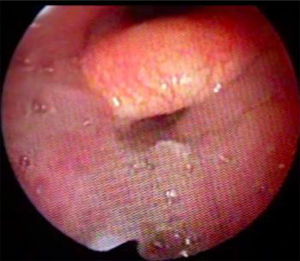
Therefore, TORS was offered only to patients with tongue base hypertrophy, using the surgical technique described by Vicini et al. (18), which consists mainly of complete removal of the lingual tonsil with a minimal tongue resection, performed using the Da Vinci® surgical system for TORS. This procedure was combined with a functional expansion pharyngoplasty, also performed with the Da Vinci® system, offering a multilevel treatment (19).
The postoperative PSG was performed on average six months after surgery. The treatment was considered effective if the postoperative AHI reached values below 15 events/hour, along with at least 50% reduction from the baseline AHI.
Statistical analysis was performed using SPSS (IBM Corp. Released 2015. IBM SPSS Statistics for Mac, Version 23. Armonk, NY: IMB Corp.). A Wilcoxon paired signed-rank test was used to compare preoperative and postoperative AHI and minimal oxygen saturation. The difference was considered statistically significant if the P value was less than 0.05.
Results
After clinical evaluation, 146 patients were included in this study to be submitted to DISE as candidates for surgical treatment. Lingual tonsil hypertrophy was classified according Friedman scoring system as grade 1 and 2 in 12% of the evaluated cases and as grade 3 and 4 in other 12%. During DISE, retroglossal obstruction was detected in 109 patients (74%) (Figure 4).
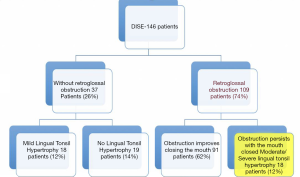
Following the selection criteria proposed for this study, TORS was offered to only 18 patients, in which tongue base obstruction persisted after the maneuver, confirming that the obstruction was caused exclusively by the lingual tonsils hypertrophy (grade 3 and 4). Patients with palatine tonsils hypertrophy (grade 3 and 4) were previously excluded.
Finally, the selected group was 89% male with average age 38±9.3 years and BMI 29.8±4.4 kg/m2. The mean baseline AHI was 32±15.4 events/hour (Figure 5).
The subjects included in this study were submitted to the same procedure, TORS for TBR associated with a functional expansional pharyngoplasty, since besides the tongue base obstruction all had presented during DISE obstruction at the velum and 67% had also oropharyngeal obstruction. The collapse at the epiglottis level was seen in 27% of the patients, but always secondary to the tongue base obstruction and partial epiglottectomy was not performed in any patient.
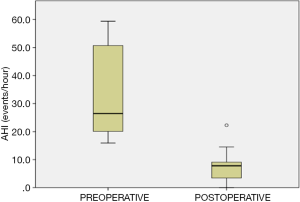
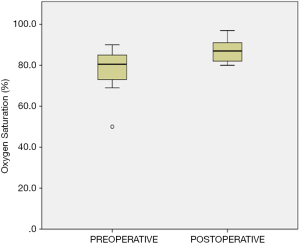
The postoperative AHI was 7.3±5.1 events/hour and 88.9% of the subjects met the criteria for surgical success. There was a significant statistical difference between baseline and postoperative AHI (P=0.0001) (Figures 6 and 7). The minimum oxygen saturation increased from 78±9.6 to 87±5.9 (P=0.0001) (Figure 8).
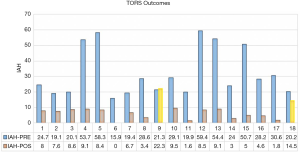
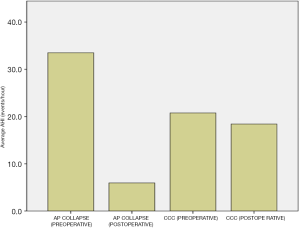
Patients with palatal AP collapse had the most significant improvement, the AHI reduced from 33.5±15.8 to 5.9±3.1 (P=0.0001). Patients with palatal circumferential collapse (CC) did not have the same reduction in AHI, as baseline AHI was 20.7±0.5 and postoperative AHI was 18.4±3.9 (P=0.15). There was a statistically significant difference of the postoperative AHI between patients with and without palatal CC (P=0.001) (Figure 9).
The average lingual tissue volume removed was 11±4 cc and no patient experienced severe complications after the procedure. We did not observe changes in BMI during the studied period.
Discussion
This study evaluates DISE as a tool to select candidates to TORS for OSA surgical treatment. The results suggest that the adopted protocol was useful in identifying when the tongue base collapse was really secondary to tongue base hypertrophy, contributing to achieve good surgical outcomes by excluding patients with lack of muscular support for the tongue.
Furthermore, our data have demonstrated that the presence of complete circumferential collapse (CCC) at the velum may predict a worse surgical outcome even when a multilevel procedure is performed.
Persistence of CCC at the velum, lateral pharyngeal wall and tongue base have been described in several studies using DISE to evaluate non-responders to UPPP (20), suggesting that this procedure was not able to address all these obstructive sites.
Focusing on the treatment of the lateral pharyngeal wall collapse, one of the possible causes of UPPP failure, other pharyngeal surgeries were described in the last decade, achieving an increase in success rate to values up to 65% (21-23). Nevertheless, a high percentage of patients did not responsive favorably to this treatment, which can be explained by the fact that this procedure is not addressing properly the retroglossal obstruction.
The TORS was first described in OSA surgical treatment precisely to treat the tongue base and supraglottic collapse (23), using the Da Vinci® surgical system, a technological platform initially developed for the treatment of oropharyngeal cancer.
TORS in association with ESP has achieved a success rate up to 75% in OSA surgical treatment, result attributed to the multilevel approach, offering in the same procedure a proper treatment to all possible obstructive sites (24).
However, systematic reviews of TBR for OSA treatment show varying results (25,26), which might be consequence of the patient selection criteria adopted in each study. Performing the same TORS technique to treat patients with tongue base obstruction and adopting the same success criteria, defined as postoperative AHI under 20 events/hour and AHI reduction of at least 50% from baseline, Vicini et al. obtained a success rate of 70% (27), Lee et al. a success rate of 45% (28) and Lin et al achieved good surgical response in 53.8% of cases (29).
Ultimately, DISE has been used by many centers around the world as a tool for evaluating candidates to tongue base and hypopharyngeal surgery in OSA surgical treatment, but the lack of standardization on its technique and interpretation may explain why patient selection using DISE did not improve the surgical outcomes (30).
We advocate that there is often a misinterpretation regarding the diagnose of tongue base and hypopharyngeal obstruction detected by DISE, taking into account only the obstructive site to propose the surgical procedure, ignoring the underlying mechanisms causing the upper airway collapse. Theoretically, if the lack of muscular support leads the tongue to collapse, removing soft tissue will not provide a good surgical outcome.
In our study, we assumed the hypothesis that DISE can be interpreted in such a way as to identify the tongue base collapse due to lack of muscular support, detecting with better accuracy patients with tissue hypertrophy. It may explain our success rate up to 90%, once we were able to contraindicate the procedure in potential non-responders.
Other studies have demonstrated the absence of velopharyngeal lateral collapse, BMI <30, AHI <60 and no previous pharyngeal surgeries as predictor factors to better TORS surgical outcome for OSA treatment (31,32), characteristics also present in the population selected in this study, even though it was not a selection criteria adopted.
Analyzing our two surgical failures and comparing with the group who have achieved good surgical outcomes, the only difference was the presence of palatal circumferential collapse in preoperative DISE. Both patients were submitted to a postoperative DISE for further comprehension and only the CC at the velum was still present, data corroborated by other study in which the gain in velopharyngeal area was also correlated with AIH reduction (33), suggesting that the outcome was not compromised by the ability of TORS in improving the oropharyngeal and hypopharyngeal areas.
It is increasingly evident in literature that the anatomy is not the only determinant variable in OSA physiopathology and factors affecting the muscular support for the tongue seems to have a paramount role in OSA physiopathology (33).
Thus, the use of DISE as a tool to detect the tongue base and hypopharyngeal obstruction lead by the lack of muscular support might promote a positive impact in the surgical outcomes. This concept was previously tested measuring preoperatively the genioglossus activity with electromyography, which was demonstrated to be also effective (33)
Although the present study have some methodological limitations, was a retrospective analysis of small series of cases without randomization, we were able to demonstrate a significant statistical difference between preoperative and postoperative AHI, suggesting that this concept seems to be reliable to evaluate the tongue base collapse in patient selection to TORS in OSA treatment.
Conclusions
In the present study, the obtained results highlight the importance of differentiating tongue base obstruction due to soft tissue hypertrophy from the lack of muscular support, reinforcing the concept that DISE criteria adopted to select patients for TORS might be able to influence the surgical outcomes. We emphasize the need of further investigation including a control group to prove this concept as a way to improve TORS surgical outcomes.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ajo-19-68). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study protocol was approved by the Institutional Review Board of Hospital Samaritano - SP (52479116.3.0000.5487) and was conducted in accordance with the Helsinki Declaration (as revised in 2013). The authors confirm that written informed consent was dismissed by the IRB for publication (retrospective review of electronic medical record) of this article and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Tufik S, Santos-Silva R, Taddei JA, et al. Obstructive sleep apnea syndrome in the Sao Paulo Epidemiologic Sleep Study. Sleep Med 2010;11:441-6. [Crossref] [PubMed]
- Young T, Finn L, Peppard PE, et al. Sleep disordered breathing and mortality: eighteen-year follow-up of the Wisconsin sleep cohort. Sleep 2008;31:1071-8. [PubMed]
- Wang X, Ouyang Y, Wang Z, et al. Obstructive sleep apnea and risk of cardiovascular disease and all-cause mortality: a meta-analysis of prospective cohort studies. Int J Cardiol 2013;169:207-14. [Crossref] [PubMed]
- Shahar E, Whitney CW, Redline S, et al. Sleep-disordered breathing and cardiovascular disease: cross-sectional results of the Sleep Heart Health Study. Am J Respir Crit Care Med 2001;163:19-25. [Crossref] [PubMed]
- Mokhlesi B, Ayas NT. Cardiovascular Events in Obstructive Sleep Apnea - Can CPAP Therapy SAVE Lives? N Engl J Med 2016;375:994-6. [Crossref] [PubMed]
- He M, Yin G, Zhan S, et al. Long-term Efficacy of Uvulopalatopharyngoplasty among Adult Patients with Obstructive Sleep Apnea: A Systematic Review and Meta-analysis. Otolaryngol Head Neck Surg 2019;161:401-11. [Crossref] [PubMed]
- Miller SC, Nguyen SA, Ong AA, et al. Transoral robotic base of tongue reduction for obstructive sleep apnea: A systematic review and meta-analysis. Laryngoscope 2017;127:258-65. [Crossref] [PubMed]
- Justin GA, Chang ET, Camacho M, et al. Transoral Robotic Surgery for Obstructive Sleep Apnea: A Systematic Review and Meta-Analysis. Otolaryngol Head Neck Surg 2016;154:835-46. [Crossref] [PubMed]
- Friedman M, Ibrahim H, Bass L. Clinical staging for sleep-disordered breathing. Otolaryngol Head Neck Surg 2002;127:13-21. [Crossref] [PubMed]
- Friedman M, Salapatas AM, Bonzelaar LB. Updated Friedman Staging System for Obstructive Sleep Apnea. Adv Otorhinolaryngol 2017;80:41-8. [Crossref] [PubMed]
- Eckert DJ, Malhotra A. Pathophysiology of adult obstructive sleep apnea. Proc Am Thorac Soc 2008;5:144-53. [Crossref] [PubMed]
- Hoshino Y, Ayuse T, Kurata S, et al. The compensatory responses to upper airway obstruction in normal subjects under propofol anesthesia. Respir Physiol Neurobiol 2009;166:24-31. [Crossref] [PubMed]
- Hohenhorst W, Ravesloot MJL, Kezirian EJ, de Vries N. Drug-induced sleep endoscopy in adults with sleep-disordered breathing: Technique and the VOTE Classification system. Oper Tech Otolayngol Head Neck Surg 2012;23:11-8. [Crossref]
- De Vito A, Carrasco Llatas M, Ravesloot MJ, et al. European position paper on drug-induced sleep endoscopy: 2017 Update. Clin Otolaryngol 2018;43:1541-52. [Crossref] [PubMed]
- Viana AC, Thuler LC, Araújo-Melo MH. Drug-induced sleep endoscopy in the identification of obstruction sites in patients with obstructive sleep apnea: a systematic review. Braz J Otorhinolaryngol 2015;81:439-46. [Crossref] [PubMed]
- Patil SP, Schneider H, Marx JJ, et al. Neuromechanical control of upper airway patency during sleep. J Appl Physiol 1985;2007:547-56. [PubMed]
- Rabelo FA, Küpper DS, Sander HH, et al. Polysomnographic evaluation of propofol-induced sleep in patients with respiratory sleep disorders and controls. Laryngoscope 2013;123:2300-5. [Crossref] [PubMed]
- Vicini C, Montevecchi F, Gobbi R, et al. Transoral robotic surgery for obstructive sleep apnea syndrome: Principles and technique. World J Otorhinolaryngol Head Neck Surg 2017;3:97-100. [Crossref] [PubMed]
- Sorrenti G, Piccin O. Functional expansion pharyngoplasty in the treatment of obstructive sleep apnea. Laryngoscope 2013;123:2905-8. [Crossref] [PubMed]
- Green KK, Kent DT, D'Agostino MA, et al. Drug-Induced Sleep Endoscopy and Surgical Outcomes: A Multicenter Cohort Study. Laryngoscope 2019;129:761-70. [Crossref] [PubMed]
- Cahali MB. Lateral pharyngoplasty: a new treatment for obstructive sleep apnea hypopnea syndrome. Laryngoscope 2003;113:1961-8. [Crossref] [PubMed]
- Pang KP, Pang EB, Win MT, et al. Expansion sphincter pharyngoplasty for the treatment of OSA: a systemic review and meta-analysis. Eur Arch Otorhinolaryngol 2016;273:2329-33. [Crossref] [PubMed]
- Vicini C, Montevecchi F, Campanini A, et al. Clinical outcomes and complications associated with TORS for OSAHS: a benchmark for evaluating an emerging surgical technology in a targeted application for benign disease. ORL J Otorhinolaryngol Relat Spec 2014;76:63-9. [Crossref] [PubMed]
- Vicini C, Montevecchi F, Magnuson JS. Robotic Surgery for Obstructive Sleep Apnea. Curr Otorhinolaryngol Rep 2013;1:130-6. [Crossref] [PubMed]
- Meccariello G, Cammaroto G, Montevecchi F, et al. Transoral robotic surgery for the management of obstructive sleep apnea: a systematic review and meta-analysis. Eur Arch Otorhinolaryngol 2017;274:647-53. [Crossref] [PubMed]
- Thaler ER, Rassekh CH, Lee JM, et al. Outcomes for multilevel surgery for sleep apnea: Obstructive sleep apnea, transoral robotic surgery, and uvulopalatopharyngoplasty. Laryngoscope 2016;126:266-9. [Crossref] [PubMed]
- Vicini C, Dallan I, Canzi P, et al. Transoralrobotic surgery of the tongue base in obstructive sleep Apnea-Hypopnea syndrome:anatomic considerations and clinical experience. Head Neck 2012;34:15-22. [Crossref] [PubMed]
- Lee JM, Weinstein GS, O'Malley BW Jr, et al. Transoral robot-assistedlingual tonsillectomy anduvulopalatopharyngoplasty for obstructive sleep apnea. Ann Otol Rhinol Laryngol 2012;121:635-9. [Crossref] [PubMed]
- Lin HS, Rowley JA, Folbe AJ, et al. Transoral robotic surgery for treatment of obstructive sleep apnea: factors predicting surgical response. Laryngoscope 2015;125:1013-20. [Crossref] [PubMed]
- Chiffer RC, Schwab RJ, Keenan BT, et al. Volumetric MRI analysis pre- and post-Transoral robotic surgery for obstructive sleep apnea. Laryngoscope 2015;125:1988-95. [Crossref] [PubMed]
- Eckert DJ, White DP, Jordan AS, et al. Defining phenotypic causes of obstructive sleep apnea. Identification of novel therapeutic targets. Am J Respir Crit Care Med 2013;188:996-1004. [Crossref] [PubMed]
- Eckert DJ. Phenotypic approaches to obstructive sleep apnoea - New pathways for targeted therapy. Sleep Med Rev 2018;37:45-59. [Crossref] [PubMed]
- Zhao D, Li Y, Xian J, et al. The Combination of Anatomy and Genioglossus Activity in Predicting the Outcomes of Velopharyngeal Surgery. Otolaryngol Head Neck Surg 2017;156:567-74. [Crossref] [PubMed]
Cite this article as: Thuler E, dos Santos Junior V, Yui MS, Tominaga Q, Rabelo FAW. Correlation between drug-induced sleep endoscopy criteria for patient selection and transoral robotic surgery outcomes in obstructive sleep apnea treatment. Aust J Otolaryngol 2020;3:20.



