Endoscopic myringoplasty, a retrospective cohort study
Introduction
Chronic suppurative otitis media (CSOM) is a well-described condition characterised by inflammation of the middle ear associated with infection progressing to ear drum perforation with purulent discharge (1). One of the most common complications of CSOM is hearing loss, underlying the importance of optimal definitive treatment (2). Otitis media-related hearing impairment has a prevalence of 30.8 per 10,000 people worldwide, with as many as 21,000 individuals dying each year due to complications (3). Otitis media and perforation of the tympanic membrane (TM) tend to occur concurrently. The intended outcome of treating complications of otitis media, such as TM perforation, is for the insult to heal spontaneously; however, this is not always the case. When longstanding chronic perforations persist treatment focuses on the improvement of conductive hearing loss and repair of the anatomical defect, which is performed by closure through application of a graft. This is better known as myringoplasty, one of the most common procedures performed in otolaryngology today. Whilst CSOM is a common cause for TM perforation, other aetiologies do exist, including, but not limited to: head trauma, barotrauma, iatrogenic perforation and complications of otitis externa. Microscopes and endoscopes alike have both played a role in the treatment of middle ear pathology, with the former remaining the preferred viewing approach due to availability and cost. As such, the implementation of surgical teaching still occurs by microscope.
Endoscope-assisted ear surgery has increased in popularity since it was first observed as a diagnostic tool in 1967 (4). Thereafter, it surged in practical application in the late 1990s, but is yet to show a definitive correlation with improved success when compared to the traditional microscopic ear surgery method, notably in TM repair by myringoplasty (5,6). Noted experiences focusing on different global regions and age groups continue to show no clear winner (7,8). Generally, the feature that defines both methods is the surgical view. While microscope-assisted myringoplasty (MAM) does allow for binocular vision and two-handed operating, it is limited by the narrowest portion of the external auditory canal. In endoscope-assisted myringoplasty (EAM), the endoscope can advance past this section, obtaining a much wider and clearer image of the surgical field from its angled lens at the distal end of the instrument, as well as heightened image resolution. This creates the potential of avoiding an invasive operative approach (such as postauricular incision and canaloplasty) to achieve successful grafting. Therefore, the endoscopic approach affords improved cosmesis, reduced intraoperative bleeding and reduced postoperative pain (9).
So far, the literature has focused on the advantages and disadvantages of both methods and less on the outcomes of graft success and audiometry. In light of this, the following retrospective study examines a 5-year period of both microscopic and endoscopic myringoplasty performed at a peripheral hospital in Australia with an otology service. This study also compares graft success rates and changes in pre and postoperative three-frequency, pure-tone average (PTA) audiometry, and whether the endoscopic assisted over microscopic approach offers greater advantage.
We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/ajo-20-56).
Methods
This cohort study was carried out retrospectively by comparing medical records of all patients who have undergone endoscopic or microscopic ear surgery at Logan Public Hospital in Queensland, Australia, from June 2013 to June 2018. Research was conducted using a combination of electronic and paper records detailing patients’ clinical and pathological history (n=87), as provided with their consent. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was reviewed and approved by the Metro South Health Human Research Ethics Committee (approval number LNR/2019/QMS/53002) and was deemed of negligible risk in accordance with the National Statement on Ethical Conduct in Human Research by the National Health and Medical Research Council of Australia. All patients enrolled completed the informed consent form. Site-specific assessment was also obtained, outlining no financial costs associated with the research. The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The outcomes of this study will not affect future management of the patients.
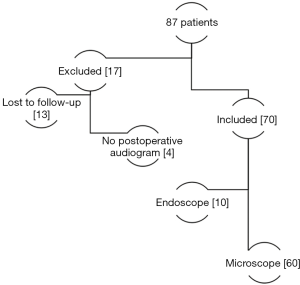
All patients with a history of cholesteatoma or previous ear surgery were excluded from this study, as were patients undergoing combined operations (e.g., ossiculoplasty). In total, 70 of 87 patients aged between 4 and 70 years (53% females and 47% males) met the inclusion criteria (Figure 1). Seventeen patients were excluded due to lack of postoperative audiogram (n=4) and loss to follow-up (n=13).
The data obtained include age, gender, surgical approach, visualisation method and graft type. In addition, types of myringoplasty surgical approach such as endaural, transcanal, postauricular and endaural plus post auricular were recorded. Also charted were graft types such as fat, tragal cartilage, conchal bowl cartilage, triangular fossa cartilage, temporalis fascia and conchal bowl cartilage plus temporalis fascia.
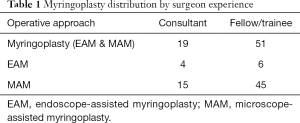
Patients of any TM defect size were considered for analysis. A total of 60 patients underwent MAM (n=60) and 10 patients underwent EAM (n=10). EAM was defined as a combination of endoscope-microscope use. MAM was defined as solely microscope use. Of the 70 operations, 19 (27%) were performed by a consultant surgeon and 51 (73%) by an otolaryngology trainee or fellow. Consultant surgeons performed 4 EAM and 15 MAM. Fellows or trainees performed 6 EAM and 45 MAM (Table 1). Surgical failure was determined to occur with persistent graft failure between 6 and 24 weeks. Patients then underwent routine pre- and postoperative audiometry, with PTA calculated thereafter at 500, 100 and 2,000 Hz. Changes to PTA were derived from difference in the pre and postoperative values in decibels (dB).
Full table
Statistical analysis was performed using R Statistical Software (version 3.5.3, R Foundation for Statistical Computing, Vienna, Austria). Also conducted was the Fisher’s exact test, to assess the difference between graft failure rate in EAM and MAM, stratifying for level of surgeon experience, and the Welch two-sample t-test, to measure any improvement in PTA between the two methods, again stratifying for surgeon experience.
Results
Seventy patients with persistent TM perforation requiring surgical repair by myringoplasty were included for study. The sample was divided into two groups according to surgical approach: MAM (n=60) and EAM (n=10). There were 37 females and 33 males. The operation was performed on the left ear for 34 patients and on the right ear for 36 patients. The mean patient age [± standard deviation (SD)] was 37.87±19.27 years.
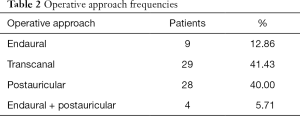
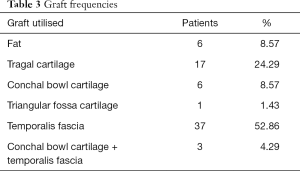
Four operative approaches to myringoplasty (utilising either microscope or combination of endoscope and microscope) were performed, the most common of which being transcanal myringoplasty (41.43%) (Table 2). Graft material was then harvested from five locations around the ear, notably from the temporalis fascia (52.86%) (Table 3).
Full table
Full table
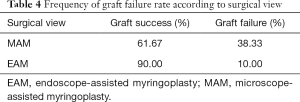
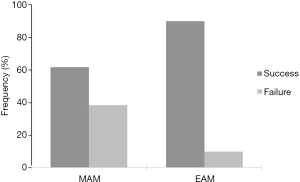
Approximately 34% of all procedures experienced persistent graft failure from 6 weeks to 12 months or more. However, the MAM failure rate (38.33%) was almost four times as high as EAM failure (10%) (Fisher’s exact test: P=0.15; 95% confidence interval (CI): 0.00, 1.47; odds ratio 0.18) (Table 4, Figure 2). Results were stratified based on the level of experience of the operative surgeon to account for this significant confounding variable. Trainees and fellows experienced a MAM failure rate of 40% and an EAM failure rate of 16.7% (P=0.39) whilst consultants suffered a MAM failure rate of 33.3% and an EAM failure rate of 0% (P=0.53).
Full table
Across all patients, the mean preoperative PTA (± SD) was 42.40±18.07 dB, and the postoperative PTA (± SD) was 33.46±19.01 dB. The change in PTA for the EAM group was on average 5 dB lower than the MAM group and had less variability by comparison. SDs of 13.80 and 6.40 for the MAM and EAM, respectively, were also noted. The mean change in MAM PTA ± SD was –8.24±13.8 dB, and the mean change for the EAM PTA ± SD was –13.20±6.40 dB. The mean change in difference in PTA between pre- and postoperative audiometry (± SD) was –8.94±13.1 dB (Welch two-sample t-test: P=0.08; 95% CI: –0.62, 10.4) (Table 5). Results were stratified for level of surgeon experience. Consultants demonstrated an average improvement in PTA of 11 dB using EAM compared to the MAM (Welch two sample t-test: P=0.006) (Table 6). Trainees and fellows demonstrated an average improvement in PTA of 2 dB using EAM compared to MAM (Welch two sample t-test: P=0.62) (Table 7).

Full table

Full table

Full table
Discussion
Minimally invasive surgery has undergone significant developments over the last 30 years, with current evidence supporting its widespread adoption due to a reduced perioperative stress response associated with increased metabolic demand, catabolism and decreased inflammatory response to surgery (among other physiological changes) (10). Pursuit of this has, within the field of otolaryngology, led to procedures similar to EAM. The current literature reflects a breadth of other experimental uses for the endoscope in middle-ear surgery, including cochlear implantation (11).
The transcanal endoscopic approach transforms the external auditory canal into a surgical portal, allowing less-invasive surgery with reduced need for a postaural incision and canaloplasty, as well as better postoperative recovery. This is measured by a return to normal daily activity and patient discharge, in addition to better cosmesis (12). In paediatric patients, the endoscopic approach can be beneficial in more challenging cases exhibiting a narrow ear canal (13). That said, the benefits also have accompanying shortcomings. For example, loss of depth perception with monocular vision when switching from endoscope to microscope can be detrimental to surgical skill. In addition to this, one-handed operating must be performed while the acting surgeon’s second hand controls the endoscope, possibly affecting operation time.
This study also aimed to compare graft success rates and changes in pre and postoperative PTA, including whether there is greater advantage of EAM over MAM. To narrow the study scope, we chose to exclude combined procedures such as the addition of ossiculoplasty to ensure homogeneity between groups. As previous research shows, more complicated procedures are less likely to result in hearing improvement (14).
Previous literature also varies significantly on the graft failure rates of myringoplasty (15-19). This study showed persistent graft failure in 34.29% of patients post-myringoplasty, irrespective of surgical approach, visualisation method or level of surgeon experience. Factors affecting graft success also vary greatly, with Albu et al. (20) highlighting in the literature that failure rates can be attributed to various factors such as smoking status of patients, a healthy opposite ear, and a long dry period preceding the operation.
Numbers of EAM were limited in this study, despite this, a trend towards significance is noted in the failure rate. Overall, the quoted MAM failure rate (38.33%) in this study is almost four times as high as the EAM (10%). Stratification for surgeon experience also demonstrates a near four-fold improvement in graft failure rate in both groups. The lower EAM failure rate is likely attributed to improved vision of the TM during graft placement. Further, the time period used to define persistent graft failure may have also confounded our reported failure rate. A more controlled definition could benefit from future research, as previous literature also lacks a definitive standard of graft failure (15-19).
The study further noted that improved function of hearing is a direct measure of quality of life following ear surgery. Changes in audiometry provide an analytical correlate in patients with complaints of hearing loss. When comparing EAM to MAM, the results showed a discrepancy in the latter group, with PTA 5 dB lower as well as decreased variability. Of importance, consultant surgeons demonstrated a statistically significant improvement in hearing thresholds in the EAM compared to the MAM groups. This has been noted by Kuo and Wu (21) also found that EAM improved hearing outcomes as a significant result. Although the mean change in difference between PTA pre and postoperatively for consultant lead myringoplasty was significant (P=0.006), a strong conclusion cannot be made at this stage due to limited number of EAMs performed. Other factors that have shown to affect hearing improvement with graft success include perforation size, with greater hearing improvement expected in perforations involving >50% of the TM.
Size also matters when investigating graft failure. In the literature, Lee et al. (14) reported a 74.1% graft success rate in perforations approximated to be <50% of the TM pars tensa; this dropped to 56% when approximated to be ≥50% perforation (14). As such, future research should focus on the presence of PTA discrepancy proportionality to membrane perforation size, in addition to controlling for the anatomical variation to the operation that is performed and the graft type utilised. EAM is also currently disadvantaged by a one-handed surgical technique. Recently, a novel concept was proposed—which sees the endoscope attached to a plate to secure it in place for two-handed endoscopic surgery (22)—but this requires a prospective randomised control trial measured against traditional two-handed MAM operating to establish a more level playing field when comparing both surgical approaches.
Overall, endoscopes provide easier navigation through the ear canal as well as improved observation with a magnified view. Despite its disadvantages, it is a serious contender in providing better outcomes than traditional microscope-assisted ear surgery. This study compared microscopic and endoscopic myringoplasty to determine whether endoscopic methods improve both graft success rate and hearing. The findings showed persistent graft failure in 34.29% of patients post-myringoplasty, irrespective of surgical approach with a near four-fold increase in MAM compared to EAM irrespective of surgeon experience. As a result, the MAM group recorded PTA levels 5 dB lower than EAM, with less variability. Also noted was a significant mean change between PTA pre- and postoperatively. Furthermore, hearing thresholds were significantly improved when using EAM in the hands of a consultant otolaryngology, head and neck surgeon. Overall, the study suggests that endoscopes offer an effective alternative for microscope-assisted ear surgery and should be further investigated in more controlled conditions, particularly as two-handed endoscopic operating is now a possibility.
Acknowledgments
The authors would like to thank Associate Professor Bernard Whitfield and the Department of Otolaryngology at Logan Hospital, Queensland. They acknowledge the Metro South Health Centres for Health Research for facilitation of the Metro South Health Biostatistics Service provided by the Queensland Facility for Advanced Bioinformatics and funded by the Metro South Study, Education and Research Trust.
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/ajo-20-56
Data Sharing Statement: Available at http://dx.doi.org/10.21037/ajo-20-56
Peer Review File: Available at http://dx.doi.org/10.21037/ajo-20-56
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ajo-20-56.). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was reviewed and approved by the Metro South Health Human Research Ethics Committee (approval number LNR/2019/QMS/53002) and was deemed of negligible risk in accordance with the National Statement on Ethical Conduct in Human Research by the National Health and Medical Research Council of Australia.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Harkness P, Topham J. Classification of otitis media. Laryngoscope 1998;108:1539-43. [Crossref] [PubMed]
- Aarhus L, Tambs K, Kvestad E, et al. Childhood otitis media: A cohort study with 30-year follow-up of hearing (the HUNT study). Ear Hear 2015;36:302-8. [Crossref] [PubMed]
- Monasta L, Ronfani L, Marchetti F, et al. Burden of disease caused by otitis media: Systematic review and global estimates. PloS One 2012;7:e36226 [Crossref] [PubMed]
- Mer SB, Derbyshire AJ, Brushenko A, et al. Fiberoptic endotoscopes for examining the middle ear. Arch Otolaryngol 1967;85:387-93. [Crossref] [PubMed]
- Tseng CC, Lai MT, Wu CC, et al. Comparison of the efficacy of endoscopic tympanoplasty and microscopic tympanoplasty: A systematic review and meta-analysis. Laryngoscope 2017;127:1890-6. [Crossref] [PubMed]
- Jyothi AC, Shrikrishna BH, Kulkarni NH, et al. Endoscopic myringoplasty versus microscopic myringoplasty in tubotympanic CSOM: A comparative study of 120 cases. Indian J Otolaryngol Head Neck Surg 2017;69:357-62. [Crossref] [PubMed]
- Yiannakis CP, Sproat R, Iyer A. Preliminary outcomes of endoscopic middle-ear surgery in 103 cases: A UK experience. J Laryngol Otol 2018;132:493-6. [Crossref] [PubMed]
- Han SY, Lee DY, Chung J, et al. Comparison of endoscopic and microscopic ear surgery in pediatric patients: A meta-analysis. Laryngoscope 2019;129:1444-52. [Crossref] [PubMed]
- Harugop AS, Mudhol RS, Godhi RA. A comparative study of endoscope assisted myringoplasty and micrsoscope assisted myringoplasty. Indian J Otolaryngol Head Neck Surg 2008;60:298-302. [Crossref] [PubMed]
- Pache B, Hübner M, Jurt J, et al. Minimally invasive surgery and enhanced recovery after surgery: The ideal combination? J Surg Oncol 2017;116:613-6. [Crossref] [PubMed]
- Kozin ED, Gulati S, Kaplan AB, et al. Systematic review of outcomes following observational and operative endoscopic middle ear surgery. Laryngoscope 2015;125:1205-14. [Crossref] [PubMed]
- De Zinis LO, Berlucchi M, Nassif N. Double-handed endoscopic myringoplasty with a holding system in children: Preliminary observations. Int J Pediatr Otorhinolaryngol 2017;96:127-30. [Crossref] [PubMed]
- Tseng CC, Lai MT, Wu CC, et al. Endoscopic transcanal myringoplasty for anterior perforations of the tympanic membrane. JAMA Otolaryngol Head Neck Surg 2016;142:1088-93. [Crossref] [PubMed]
- Lee P, Kelly G, Mills RP. Myringoplasty: Does the size of the perforation matter? Clin Otolaryngol Allied Sci 2002;27:331-4. [Crossref] [PubMed]
- Biswas SS, Hossain MA, Alam MM, et al. Hearing evaluation after myringoplasty. Bangladesh J Otorhinolaryngol 2010;16:23-8. [Crossref]
- Das A, Sen B, Ghosh D, et al. Myringoplasty: Impact of size and site of perforation on the success rate. Indian J Otolaryngol Head Neck Surg 2015;67:185-9. [Crossref] [PubMed]
- El-Hennawi DEM, Ahmed MR, Abou-Halawa AS, et al. Endoscopic push-through technique compared to microscopic underlay myringoplasty in anterior tympanic membrane perforations. J Laryngol Otol 2018;132:509-13. [Crossref] [PubMed]
- Carr SD, Strachan DR, Raine CH. Factors affecting myringoplasty success. J Laryngol Otol 2015;129:23-6. [Crossref] [PubMed]
- Kamath MP, Sreedharan S, Rao AR, et al. Success of myringoplasty: Our experience. Indian J Otolaryngol Head Neck Surg 2013;65:358-62. [Crossref] [PubMed]
- Albu S, Trabalzini F, Amadori M. Usefulness of cortical mastoidectomy in myringoplasty. Otol Neurotol 2012;33:604-9. [Crossref] [PubMed]
- Kuo CH, Wu HM. Comparison of endoscopic and microscopic tympanoplasty. Eur Arch Otorhinolaryngol 2017;274:2727-32. [Crossref] [PubMed]
- Khan MM, Parab SR. Novel concept of attaching endoscope holder to microscope for two handed endoscopic tympanoplasty. Indian J Otolaryngol Head Neck Surg 2016;68:230-40. [Crossref] [PubMed]
Cite this article as: Lynch A, See M, Chang A. Endoscopic myringoplasty, a retrospective cohort study. Aust J Otolaryngol 2021;4:4.