
Reduced mean baseline impedance aids diagnosis of laryngopharyngeal reflux and gastroesophageal reflux disease
Introduction
Multichannel intraluminal impedance (MII) monitoring is currently used to investigate gastroesophageal reflux disease (GERD). Electrical impedance is measured across multiple paired electrodes mounted along a naso-esophageal catheter, and detects liquid and gas reflux boluses as changes in impedance travelling in antegrade fashion. Liquid, regardless of acidity, will be detected as a momentary reduction in impedance, since liquid acts as an excellent conductor. Different to this is the baseline impedance of the resting state of the catheter against oesophageal mucosa Between reflux events and swallows, the esophageal lumen is collapsed, and catheter contact with the esophageal wall provides a measurable baseline impedance of the esophageal mucosa.
Prolonged acid exposure has been shown to reduce baseline mucosal impedance in both animals and humans (1). Kessing et al. (2) demonstrated significantly lower baseline impedance levels in GERD patients with both pathological and physiological acid exposure times (AET) compared with healthy controls. AET has repeatedly been negatively correlated with distal baseline impedance, further demonstrating that baseline impedance is reduced by persistent acid exposure (2-4).
Mean nocturnal baseline impedance (MNBI) is a recently described measure of the baseline impedance of three 10-minute periods during nocturnal recumbence, when tracings are less affected by swallows and refluxes (3). MNBI has been shown to increase diagnostic yield in patients with GERD (5), distinguish GERD patients who respond to proton pump inhibitors (PPIs) from those with functional heartburn, that is: patients with symptoms of GERD with negative endoscopic evidence of esophagitis, negative impedance findings, and negative symptom correlation (3), and predict improvements in symptomatic severity with treatment (4).
Laryngopharyngeal reflux (LPR) refers to the reflux of gastric contents to the larynx and pharynx causing extra-esophageal symptoms such as dysphonia, globus, and chronic cough, as well as observable signs on laryngoscopic examination such as mucosal edema (6). Current catheter-based investigations have limitations especially in measuring pharyngeal and proximal esophageal impedance. Normative reflux values for pharyngeal and proximal esophageal channels are controversial (7), “pseudo-reflux” artefact occurs due to drying or loss of catheter contact from pharyngeal mucosa (8), and inter-observer reliability in interpreting impedance results for pharyngeal reflux events is poor (9). Also, pharyngeal pH measurement (Restech) measures only oropharyngeal acidity, and is less sensitive than combined impedance-pH and with lesser symptom association probability (10). Modified reflux scintigraphy is a promising tool that we have shown to be sensitive in detecting immediate and delayed laryngopharyngeal contamination in LPR patients, and has been validated by our group (11,12). There is a need for better diagnostic tools that can accurately diagnose and assess laryngopharyngeal symptoms of reflux.
There are few studies in the literature that assess MNBI in patients with LPR. There are also few studies that assess impedance in channels along the pharynx, proximal esophagus, and distal esophagus in LPR patients. The aim of the present study was to describe MNBI findings in the pharynx, proximal esophagus, and distal esophagus in a cohort of patients with LPR, and to evaluate the use of MNBI in investigating LPR. We present the following article in accordance with the STARD reporting checklist (available at https://www.theajo.com/article/view/10.21037/ajo-21-18/rc).
Methods
Patient selection and study design
A consecutive cohort of patients with symptomatic LPR and GERD with severe symptoms who had failed medical treatment were identified from a prospectively maintained database. Patients were grouped by the predominant symptom profiles into either GERD or LPR diagnostic categories.
The selection of LPR patients involved those that had a high pre-test probability of disease based on extensive clinical and radiographic findings. Firstly, patients had undergone multi-disciplinary investigation for differential diagnoses prior to referral to a diagnostic facility for consideration of treatment, for example, exclusion of asthma or chronic obstructive pulmonary disorder. Next, a standardised symptom assessment pro forma screened for the presence of symptoms of typical GERD and LPR symptoms. The senior author’s symptom assessment pro forma captured all nine symptom categories assessed in the Reflux Symptom Index (RSI) for LPR, as well as for additional symptoms of typical heartburn, oesophageal dysmotility, and delayed gastric emptying (Appendix 1). Finally, all LPR patients underwent reflux aspiration scintigraphy and were included as they had either immediate or delayed evidence of pharyngeal contamination with refluxed radio-labelled tracer. The novel scintigraphic technique that utilises digital quantification has been previously described (11,12). Direct assessment with laryngoscopy was not uniformly utilised throughout the cohort, and was hence not included in analysis.
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Data were extracted from a research database with current approval by the Sydney Local Health District Human Research Ethics Committee (reference: LNR/12CRGH/248). Patients gave written informed consent for study under the institutional ethics committee guidelines.
Manometry measurement
All patients underwent standard water-perfused manometry using a Dentsleeve, multi-channel catheter (Mui Scientific, Ontario, Canada). The anatomic positions of the upper esophageal sphincter (UES) and lower esophageal sphincter (LES) were manometrically assessed, and distance from the nares was noted to facilitate placement of impedance-pH catheters.
Impedance-pH measurement
Twenty-four-hour dual pH and impedance monitoring was performed after ceasing proton pump inhibitors for 5 days. Under topical nasal anaesthetic, a 2.3 mm diameter trans-nasal catheter (Zephyr device, Sandhill Co, Highlands Ranch, Colorado, USA) was inserted through the nose. pH sensors were positioned close to 5 and 15 cm above the LES and exactly 2 cm above the upper border of the UES, with six impedance monitoring sensors along the catheter. Ingestion of acidic beverages was restricted but no other dietary requirements were required. The catheter was connected to an external monitoring Zephyr device which stored data over the 24-hour period.
Impedance-pH data analysis
A liquid reflux episode was defined as a decrease in impedance at least 50% of baseline, beginning in the most distal impedance channel and travelling in a retrograde fashion. The uppermost channel that detected the continuing impedance drop categorised the reflux as either a distal esophageal, proximal esophageal, or a pharyngeal reflux event. A reflux episode starting in the most distal channel and ending in the pharyngeal channels was classified as a pharyngeal reflux event. Reflux episodes with pH <4 were classified as acidic, episodes with pH ≥4 were classified as non-acidic. The time period when esophageal pH <4 was divided by total monitoring time to give AET, expressed as a percentage (%). Distal AET and proximal AET were measured. The DeMeester score, a composite score that has been validated for typical GERD which includes the AET, as well as frequency and duration of reflux episodes, was also recorded (13,14).
Baseline impedance was assessed during the night-time recumbent period. Three stable 10-minute time periods (1 am, 2 am, 3 am) were selected, and the mean baseline for each 10-minute period was calculated. The mean the three values was manually calculated to obtain the MNBI for each patient. This method of calculating MNBI was performed for the most distal esophageal channel, most proximal esophageal channel, and pharyngeal channel to obtain MNBI values for distal esophagus, proximal esophagus, and pharynx, respectively. Time periods including swallows, refluxes, and pH drops were avoided. Reviewers were blinded to the clinical information of patients.
Statistical analysis
SPSS version 24.0 (IBM Corp, NY, USA) was used for statistical analysis. Data were confirmed to be normally distributed with Shapiro-Wilk test. Data are reported as mean ± standard deviation (SD). Categorical data were analysed with the chi-squared test. Continuous data were analysed with the independent t-test. Correlations between continuous variables were analysed with Pearson’s correlation coefficient. A P value <0.05 was considered statistically significant.
Results
Clinical and demographic results
One hundred and eighty-seven consecutive patients were studied, with 105 patients in the LPR group and 82 patients in the GERD group. There was a higher proportion of females in the LPR cohort than the GERD group (71.5% vs. 56.1%; P=0.047), and were older than the GERD group [mean age 58.5 (±15.1) vs. 50.7 (±16.2) years; P=0.002].
All patients in the GERD group reported heartburn as a predominant symptom. Table 1 shows the symptoms described by the LPR group. The most common extra-esophageal symptom of LPR was cough (n=79, 75.2%), followed by throat clearing (n=77, 73.3%) and dysphonia (n=68, 64.8%). Three patients (2.9%) in the LPR group described a concomitant history of typical symptoms of GERD, namely heartburn, with all three confirming the predominance of extra-esophageal symptoms. No patients in the LPR group had evidence of erosive esophagitis or hiatus hernia on endoscopy.
Table 1
| Symptoms (LPR) | N=105 (%) |
|---|---|
| Cough | 79 (75.2) |
| Throat clearing | 77 (73.3) |
| Dysphonia | 68 (64.8) |
| Mucous | 64 (61.0) |
| Globus | 63 (60.0) |
| Regurgitation to throat | 57 (54.3) |
| Sore throat | 53 (50.5) |
| Dysphagia | 41 (39.0) |
| Dyspnoea | 35 (33.3) |
| Non-viral bronchitis | 20 (19.0) |
| Laryngospasm | 19 (18.1) |
| Non-viral pneumonia | 7 (6.7) |
| Aspiration | 7 (6.7) |
| Atypical chest pain | 2 (2.0) |
LPR, laryngopharyngeal reflux.
Impedance-pH results
Table 2 shows impedance-pH results. The GERD group had a higher DeMeester score (P=0.037), longer distal AET (P<0.001), and more frequent acid reflux episodes to the distal esophagus (P=0.011) and proximal esophagus (P=0.015) than the LPR group. AET measured in the proximal esophagus was similar between the two groups (P=0.591).
Table 2
| Impedance-pH parameter | LPR (n=105) | GERD (n=82) | P value |
|---|---|---|---|
| DeMeester score | 5.5 (±13.6) | 12.1 (±28.1) | 0.037* |
| Distal AET (%) | 1.7 (±3.9) | 8.3 (±7.9) | <0.001* |
| Proximal AET (%) | 0.2 (±1.3) | 0.4 (±3.2) | 0.591 |
| MBCT (seconds) | 15.7 (±8.2) | 16 (±8.9) | 0.847 |
| Distal reflux episodes | 46.5 (±24.5) | 64.4 (±39) | <0.001* |
| Acid | 12.6 (±14.8) | 17.2 (±17.3) | 0.011* |
| Non-acid | 33.4 (±18.3) | 44.4 (36.5) | 0.067 |
| Proximal reflux episodes | 24.3 (±14.2) | 33.6 (±26.8) | 0.003* |
| Acid | 6.6 (±8.5) | 9.9 (±9.9) | 0.015* |
| Non-acid | 17.6 (±11.8) | 24 (±26) | 0.028* |
| Pharyngeal reflux episodes | 7.4 (±7.2) | 6.9 (±7.8) | 0.683 |
| Acid | 0.21 (±1.3) | 0.22 (±1.3) | 0.959 |
| Non-acid | 7.2 (±7) | 6.8 (±7.6) | 0.725 |
*, significant result. AET, acid exposure time; LPR, laryngopharyngeal reflux; GERD, gastroesophageal reflux disease; MBCT, median bolus clearance time.
MNBI results
Table 3 shows MNBI results. In the distal esophagus, MNBI was significant lower in the GERD group compared to the LPR group (1,679±914 vs. 2,109±863; P=0.001). Similarly, in the proximal esophagus, MNBI was lower in the GERD group than the LPR group (2,289±579 vs. 2,541±471; P=0.001). In the pharynx, MNBI was similar between the two groups.
Table 3
| MNBI parameter | LPR (n=105) | GERD (n=82) | P value |
|---|---|---|---|
| Distal MNBI (ohms) | 2,109 (±863) | 1,679 (±914) | 0.001* |
| Proximal MNBI (ohms) | 2,541 (±471) | 2,289 (±579) | 0.001* |
| Pharyngeal MNBI (ohms) | 2,116 (±699) | 2,133 (±770) | 0.878 |
| MNBI ratios | |||
| Pharynx:distal | 1.58 (±1.45) | 2.11 (±2.17) | 0.009* |
| Proximal:distal | 1.37 (±1.69) | 2.12 (±2.44) | 0.003* |
*, significant result. MNBI, mean nocturnal baseline impedance; LPR, laryngopharyngeal reflux; GERD, gastroesophageal reflux disease.
The ratio of MNBI at the pharynx compared to distal esophagus was >1 in both LPR and GERD groups, with the pharynx to distal esophageal MNBI ratio being significantly higher in the GERD group (P=0.009). Similarly, MNBI ratio at the proximal esophagus compared to the distal esophagus was raised in both groups, with this ratio being significantly higher in the GERD group (P=0.003).
Subgroup analyses were performed on LPR patients to investigate possible correlations of extra-esophageal symptoms with MNBI measured at three levels: distal esophageal, proximal esophageal, and pharyngeal. Patients complaining of regurgitation had a higher mean pharyngeal MNBI compared with those that did not (2,286±518 vs. 2,218±879; P=0.005). Patients complaining of dysphagia had a higher distal esophageal MNBI compared to those that did not (1,949±745 vs. 1,931±915; P=0.05). No other symptoms listed in Table 1 correlated with MNBI at any anatomical level.
Relation of MNBI and reflux events
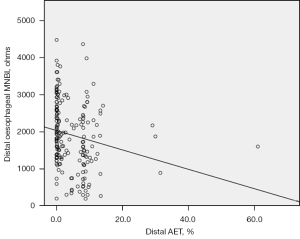
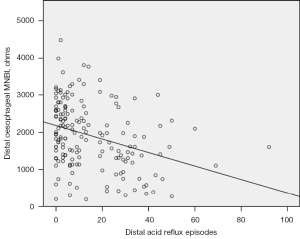
Distal AET negatively correlated with distal esophageal MNBI (r=−0.195; P=0.007) (Figure 1). The number of distal acid reflux episodes negatively correlated with distal esophageal MNBI (r=−0.330; P<0.001) (Figure 2). The number of proximal acid reflux episodes also negatively correlated with distal esophageal MNBI (r=−0.365; P<0.001), as did the number of pharyngeal acid reflux episodes (r=−0.149; P=0.042). Proximal AET did not correlate with any measurement of MNBI at any pharyngo-esophageal locations.
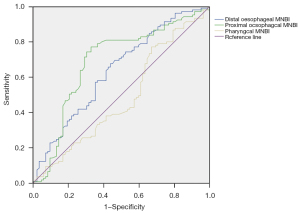
Receiver operating characteristic (ROC) curve comparing GERD and LPR
The ROC curve for MNBI measurements at the pharynx, proximal esophagus, and distal esophagus is shown in (Figure 3). The area under the ROC curves for distal esophageal MNBI was 0.638 (95% CI: 0.557–0.718; P=0.001), for proximal esophageal MNBI was 0.688 (95% CI: 0.607–0.769; P<0.001), and for pharyngeal MNBI was 0.484 (95% CI: 0.399–0.569; P=0.702)
Discussion
The present study reports MNBI values in a cohort of patients with LPR and a group of patients with treatment-refractory GERD. This is the first study to compare MNBI at three different anatomical levels between these two groups.
Both the LPR and GERD groups had reduced distal MNBI compared with reported normative values amongst healthy, asymptomatic volunteers in the literature. Reports of distal MNBI in healthy controls are 2,827 ohms by Kessing et al. (2), 3,317 ohms by Martinucci et al. (3), and 2,936 ohms by Frazzoni et al. (5). The lattermost study has established a distal MNBI value of 2,292 ohms as being the best cutoff for diagnosing GERD. The two groups of patients herein showed mean distal MNBI values under 2,292 ohms, confirming reflux abnormality. Other studies have consistently demonstrated a reduced distal MNBI in patients with pathologic AET (4), erosive reflux disease (15), and non-erosive reflux disease (16). Reduced esophageal baseline impedance hence appears to be present in both LPR and GERD, with a greater decrease in impedance in the GERD group. Decreased distal MNBI may be a useful adjunct to diagnosis in patients with appropriate symptoms.
GERD and LPR comparisons
The GERD group showed a significantly lower MNBI in the distal and proximal esophagus compared with the LPR group. Other reports that acid exposure reduces baseline impedance (1,2) are supported by the present finding that the GERD group had higher acid exposure throughout the esophagus, with significantly higher AET and more frequent acidic reflux episodes than the LPR group. Similarly, Chen and colleagues found that distal MNBI was significantly lower in patients with GERD compared with LPR patients. However, different to the present study, they report a lower proximal MNBI in patients with concomitant LPR and GERD symptoms compared with patients with pure LPR symptoms or pure GERD symptoms, despite pure GERD-symptomatic patients having the highest distal AET (17). This may suggest worse proximal mucosal damage in the combined LPR and GERD grouping. The report herein has shown higher levels of AET and episode frequency of reflux congruent with this contention. Sakin et al. also compared proximal and distal MNBI between GERD and LPR patients, and did not report a difference in distal MNBI between LPR and GERD patients (18). Interestingly, they did not report a difference in distal AET between the two groups, with both GERD and LPR groups having pathological AET (6.1% and 6.8%, respectively). It is possible that their LPR patients may have been heterogenous, with some experiencing typical reflux-like symptoms concurrently. The present study group of LPR patients largely had pure LPR symptoms (with only three patients having mixed LPR and GERD symptoms) and would seem to offer a more pure comparison between the two disease processes.
Inverse correlation between AET and MNBI
Several studies have reported an inverse correlation between MNBI and AET, indicating that acid exposure reduces baseline impedance (3,4,18). We report a similar correlation in the distal esophagus only. Both distal AET and the frequency of acid reflux episodes negatively correlated with distal MNBI. This further supports the concept that acid exposure impairs mucosal integrity, reducing baseline impedance. Dilated intercellular spaces (DIS) are a reflux-related change in mucosal cellular tight junctions. DIS has been shown to be induced by esophageal acid exposure, and accounts for the reduction in electrical impedance of mucosal tissue following prolonged acid exposure (1,19).
This study did not find that AET or the frequency of acid reflux in the proximal esophagus correlated with proximal esophageal MNBI. Congruent with the present study, Patel et al. reported that distal (but not proximal) esophageal MNBI decreased as AET values increased (4). This may be for two reasons: firstly, proximal reflux events tend to be less acidic and therefore may have a lesser effect on mucosal impairment (20). Secondly, clearance of refluxate may differ between proximal and distal esophagus, with proximal refluxate necessarily having to traverse the distal esophagus, leading to greater AET. Indeed, AET in the present cohort was far greater in the distal esophagus compared to proximal esophagus. These hypotheses are supported by the fact that baseline impedance was lowest in the distal esophagus compared to the proximal esophagus. The ratio of proximal to distal esophageal MNBI was >1 in both study groups. Proximal to distal ratio of MNBI have similarly been reported as >1 in other studies, indicating congruence in readings between authors, and a lesser degree of esophageal mucosal permeability proximally (18,21).
Symptoms analysis
When MNBI characteristics were examined by symptom profile, it was found that patients complaining of regurgitation had a higher pharyngeal baseline impedance compared with those that did not, and that patients with dysphagia had a higher distal esophageal baseline impedance compared to those that did not. However, the magnitude of difference was small, in the matter of merely 68 and 18 ohms, respectively. This is a statistically significant but clinically insignificant finding.
Pharyngeal MNBI in LPR and GERD
The utility of pharyngeal MNBI is not known. Only two studies have assessed baseline impedance in the pharynx. Doo et al. reported lower pharyngeal MNBI in LPR patients compared with healthy controls. MNBI was lowest at the pharynx, then proximal esophagus, and highest at the distal esophagus in both LPR and healthy controls (21). Dulery et al. found no difference in MNBI in the pharynx between LPR patients and healthy controls, nor did they find a difference at the proximal or distal esophagus (22). The present study reports no difference in pharyngeal MNBI between LPR and GERD groups. Although the majority of reflux episodes to the pharynx tend to be acid or weakly acid reflux (23,24), the lack of difference in pharyngeal baseline impedance may be due to pharyngeal reflux events being too brief in duration to alter mucosal integrity (22). As the pharynx is fairly capacious, integrity of baseline measurement may not be as impaired as in the distal esophagus. Indeed, pharyngeal impedance measurement may be positional, we have previously shown supine impedance measure may be advantageous in detecting impedance reflux events in the pharynx (25). The value of pharyngeal MNBI as a diagnostic tool awaits further evaluation of normal values.
Effectiveness as a diagnostic tool
The effectiveness of MNBI as a tool for diagnosing LPR was assessed by ROC analysis for three anatomical locations. The area under the receiver operating characteristic curve (AUROC) analyses for distal and proximal esophageal MNBI in our study group were fair, and for pharyngeal MNBI was poor. There are no other studies for comparison of LPR and GERD findings as yet. Frazzoni et al. report an AUROC for distal MNBI of 0.876 to diagnose GERD, which is excellent and supported by findings from other studies that baseline impedance is reduced in GERD and with symptomatic dysphagia (5,26). Pharyngeal MNBI is not currently supportable as a diagnostic test in differentiation of LPR from GERD.
Study limitations
The present study’s retrospective nature is a weakness. Baseline demographic differences between LPR and GERD groups existed. The LPR group were older and had more females than the GERD group.
Conclusions
Baseline impedance is reduced in both LPR and GERD at both distal and proximal esophageal measurements. Baseline impedance is more severely reduced in the latter group. Baseline impedance is strongly and inversely related to acid exposure in the esophagus. Distal and proximal MNBI may be useful in distinguishing LPR and GERD. Pharyngeal MNBI was not reduced or different between groups. MNBI measurement in the pharynx does not appear to be useful in differentiating LPR from GERD.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STARD reporting checklist. Available at https://www.theajo.com/article/view/10.21037/ajo-21-18/rc
Data Sharing Statement: Available at https://www.theajo.com/article/view/10.21037/ajo-21-18/dss
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://www.theajo.com/article/view/10.21037/ajo-21-18/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Data were extracted from a research database with current approval by the Sydney Local Health District Human Research Ethics Committee (reference: LNR/12CRGH/248). Patients gave written informed consent for study under the institutional ethics committee guidelines.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Farré R, Blondeau K, Clement D, et al. Evaluation of oesophageal mucosa integrity by the intraluminal impedance technique. Gut 2011;60:885-92. [Crossref] [PubMed]
- Kessing BF, Bredenoord AJ, Weijenborg PW, et al. Esophageal acid exposure decreases intraluminal baseline impedance levels. Am J Gastroenterol 2011;106:2093-7. [Crossref] [PubMed]
- Martinucci I, de Bortoli N, Savarino E, et al. Esophageal baseline impedance levels in patients with pathophysiological characteristics of functional heartburn. Neurogastroenterol Motil 2014;26:546-55. [Crossref] [PubMed]
- Patel A, Wang D, Sainani N, et al. Distal mean nocturnal baseline impedance on pH-impedance monitoring predicts reflux burden and symptomatic outcome in gastro-oesophageal reflux disease. Aliment Pharmacol Ther 2016;44:890-8. [Crossref] [PubMed]
- Frazzoni M, Savarino E, de Bortoli N, et al. Analyses of the Post-reflux Swallow-induced Peristaltic Wave Index and Nocturnal Baseline Impedance Parameters Increase the Diagnostic Yield of Impedance-pH Monitoring of Patients With Reflux Disease. Clin Gastroenterol Hepatol 2016;14:40-6. [Crossref] [PubMed]
- Koufman JA. The otolaryngologic manifestations of gastroesophageal reflux disease (GERD): a clinical investigation of 225 patients using ambulatory 24-hour pH monitoring and an experimental investigation of the role of acid and pepsin in the development of laryngeal injury. Laryngoscope 1991;101:1-78. [Crossref] [PubMed]
- Lechien JR, Akst LM, Hamdan AL, et al. Evaluation and Management of Laryngopharyngeal Reflux Disease: State of the Art Review. Otolaryngol Head Neck Surg 2019;160:762-82. [Crossref] [PubMed]
- Harrell SP, Koopman J, Woosley S, et al. Exclusion of pH artifacts is essential for hypopharyngeal pH monitoring. Laryngoscope 2007;117:470-4. [Crossref] [PubMed]
- Zerbib F, Roman S, Bruley Des Varannes S, et al. Normal values of pharyngeal and esophageal 24-hour pH impedance in individuals on and off therapy and interobserver reproducibility. Clin Gastroenterol Hepatol 2013;11:366-72. [Crossref] [PubMed]
- Ummarino D, Vandermeulen L, Roosens B, et al. Gastroesophageal reflux evaluation in patients affected by chronic cough: Restech versus multichannel intraluminal impedance/pH metry. Laryngoscope 2013;123:980-4. [Crossref] [PubMed]
- Park JS, Khoma O, Burton L, et al. A new diagnostic paradigm for laryngopharyngeal reflux disease: correlation of impedance-pH monitoring and digital reflux scintigraphy results. Eur Arch Otorhinolaryngol 2021;278:1917-26. [Crossref] [PubMed]
- Park JS, Burton L, Van der Wall H, et al. Modified Reflux Scintigraphy Detects Pulmonary Microaspiration in Severe Gastro-Esophageal and Laryngopharyngeal Reflux Disease. Lung 2021;199:139-45. [Crossref] [PubMed]
- Johnson LF, Demeester TR. Twenty-four-hour pH monitoring of the distal esophagus. A quantitative measure of gastroesophageal reflux. Am J Gastroenterol 1974;62:325-32. [PubMed]
- Neto RML, Herbella FAM, Schlottmann F, et al. Does DeMeester score still define GERD? Dis Esophagus 2019;32:doy118. [Crossref] [PubMed]
- Frazzoni M, de Bortoli N, Frazzoni L, et al. The added diagnostic value of postreflux swallow-induced peristaltic wave index and nocturnal baseline impedance in refractory reflux disease studied with on-therapy impedance-pH monitoring. Neurogastroenterol Motil 2017; [Crossref] [PubMed]
- Ribolsi M, Guarino MPL, Tullio A, et al. Post-reflux swallow-induced peristaltic wave index and mean nocturnal baseline impedance predict PPI response in GERD patients with extra esophageal symptoms. Dig Liver Dis 2020;52:173-7. [Crossref] [PubMed]
- Chen S, Liang M, Zhang M, et al. A study of proximal esophageal baseline impedance in identifying and predicting laryngopharyngeal reflux. J Gastroenterol Hepatol 2020;35:1509-14. [Crossref] [PubMed]
- Sakin YS, Vardar R, Sezgin B, et al. The diagnostic value of 24-hour ambulatory intraesophageal pH-impedance in patients with laryngopharyngeal reflux symptoms comparable with typical symptoms. United European Gastroenterol J 2017;5:632-40. [Crossref] [PubMed]
- Farré R, van Malenstein H, De Vos R, et al. Short exposure of oesophageal mucosa to bile acids, both in acidic and weakly acidic conditions, can impair mucosal integrity and provoke dilated intercellular spaces. Gut 2008;57:1366-74. [Crossref] [PubMed]
- Shay S, Tutuian R, Sifrim D, et al. Twenty-four hour ambulatory simultaneous impedance and pH monitoring: a multicenter report of normal values from 60 healthy volunteers. Am J Gastroenterol 2004;99:1037-43. [Crossref] [PubMed]
- Doo JG, Kim SI, Park JM, et al. Changes in Pharyngeal Baseline Impedance in Patients With Laryngopharyngeal Reflux. Otolaryngol Head Neck Surg 2020;163:563-8. [Crossref] [PubMed]
- Dulery C, Lechot A, Roman S, et al. A study with pharyngeal and esophageal 24-hour pH-impedance monitoring in patients with laryngopharyngeal symptoms refractory to proton pump inhibitors. Neurogastroenterol Motil 2017; [Crossref] [PubMed]
- Zerbib F, des Varannes SB, Roman S, et al. Normal values and day-to-day variability of 24-h ambulatory oesophageal impedance-pH monitoring in a Belgian-French cohort of healthy subjects. Aliment Pharmacol Ther 2005;22:1011-21. [Crossref] [PubMed]
- Hoppo T, Sanz AF, Nason KS, et al. How much pharyngeal exposure is “normal”? Normative data for laryngopharyngeal reflux events using hypopharyngeal multichannel intraluminal impedance (HMII). J Gastrointest Surg 2012;16:16-24. [Crossref] [PubMed]
- Burton L, Falk GL, Beattie J, et al. Findings from a novel scintigraphic gastroesophageal reflux study in asymptomatic volunteers. Am J Nucl Med Mol Imaging 2020;10:342-8. [PubMed]
- Cho YK. How to Interpret Esophageal Impedance pH Monitoring. J Neurogastroenterol Motil 2010;16:327-30. [Crossref] [PubMed]
Cite this article as: Park JS, Van der Wall H, Falk GL. Reduced mean baseline impedance aids diagnosis of laryngopharyngeal reflux and gastroesophageal reflux disease. Aust J Otolaryngol 2022;5:1.


