
Modified uvulopalatopharyngoplasty and radiofrequency-in-saline tongue for the management of snoring
Introduction
Snoring is a common patient (and partner) complaint, caused by upper airway vibration and turbulent airflow with a prevalence of up to 48% in the adult population (1). The majority of patients who snore are middle aged males (1). Snoring is contributed to by variations in airway shape/dimensions, low neuromuscular tone, obesity and other factors, alone or in combination (2). Snoring represents both a risk factor and cardinal symptom of obstructive sleep apnoea (OSA), a condition which carries cardiovascular and mortality risk (3). Though primary snoring without OSA has been thought to carry social rather than medical risk (3), and mild OSA [Apnoea-Hypopnea Index (AHI) between 5 and 14] is mostly only treated if symptomatic (4,5), recent literature contends that primary snoring may be a risk factor in carotid artery disease and stroke (6,7).
Snoring has a significant effect not only on the patient themselves but also on the bed partner, with studies (8) indicating bed partners lose more than an hour of sleep per night, equating to an entire night of sleep lost per week. Furthermore, it has been associated with increasing bed partner levels of daytime stress, depression and fatigue, significantly affecting quality of life (9). There is a growing need to provide an intervention to alleviate both the medical and social ramifications of this condition.
Treatment options for snoring include lifestyle measures such as weight loss and reduction in alcohol consumption, or device therapy including mandibular advancement splints, nasal expiratory positive airway pressure (EPAP) (10) and continuous positive airway pressure (CPAP) (11). Despite its effectiveness, CPAP therapy carries a substantial risk of non-compliance due to discomfort, claustrophobia, social stigma, personality factors, socio-economic factors, and healthcare beliefs/understanding (12). Practically, patients may be unwilling to wear CPAP just for snoring control, especially if the degree of OSA (e.g., AHI <15) does not confer significant risk (12). Therefore, surgical options which provide symptom relief without ongoing device use are desirable.
The soft palate is implicated as contributory in more than 90% of cases of snoring (13) yet there is a paucity of evidence assessing surgical management of snoring. Injection snoreplasty (14), palatal implants (15) and radiofrequency palatal surgery (16) have shown improvements in symptom scores in the short term. Modified uvulopalatopharyngoplasty (modUPPP) provides a reconstructive option for the soft palate, is preferred over earlier ablative techniques, and enlarges the retropalatal airway (17,18). Historically there have been various alterations of the original uvulopalatopharyngoplasty, but the procedure examined in this study is as described by Mackay and colleagues (17). The surgical technique of the modUPPP involves resection of the palatine tonsils, creation of a superolateral velopharyngeal port with removal of supratonsillar fat and transverse dissection of the posterior pharyngeal pillar; the superior part of the dissected pillar is advanced anteriorly and sutured to the previously created port, increasing the oral airway diameter (17). Radiofrequency-in-saline tongue (RIST) represents a method of tongue stiffening which creates localised plasma fields to ablate columns of tongue tissue. The modUPPP and RIST techniques each provide lower morbidity approaches compared to their respective historic counterparts to increase the oropharyngeal airway diameter and prevent airway collapse (17). Recently, the Sleep Apnea Multilevel Surgery (SAMS) multicentre clinical trial provided randomized evidence of its efficacy and effectiveness in patients with moderate or severe OSA, significantly improving both AHI and Epworth Sleepiness Scale (ESS) at 6 months (18). Furthermore, there was a significant improvement in secondary and exploratory outcomes, including snoring severity scale (SSS) and partner reported outcomes (18).
The purpose of this study is to specifically assess the effectiveness of modUPPP and radiofrequency in saline tongue surgery in the management of snoring without clinically significant OSA, defined herein by an AHI ≤15 on polysomnogram and presentation with snoring as the major concern. We present the following article in accordance with the STROBE reporting checklist (available at https://www.theajo.com/article/view/10.21037/ajo-21-47/rc).
Methods
This is a retrospective case series of prospectively gathered data. The patient population included consecutive adult patients with primary complaint snoring, and an AHI ≤15, who underwent modUPPP and RIST performed with a standardised approach (17) at a single centre in New South Wales between 2008 and 2020. All patients had previously failed device therapy (19). Twenty-five patients in the cohort also underwent nasal surgery and had prior nasal surgery based on either symptoms of nasal obstruction or as an adjunctive procedure to facilitate CPAP/mandibular advancement device (MAD) therapy use.
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the human research ethics committee attached to our local health district (ISLHD Low & Negligible Risk (LNR) Research Review Committee). All patients provided written informed consent prior to surgical intervention. Informed consent was not required for participation in this study, as advised by the ethics committee, given this was a retrospective study based on existing data.
Patients included in this study:
- Were age ≥18 years at time of modUPPP and RIST surgery;
- Had level 1 or level 2 polysomnogram conducted prior to surgery, with AHI ≤15;
- Completed the SSS questionnaire prior to, and at 3 months following surgical intervention;
- Had trialled and failed (or refused) device therapy;
- Had body mass index (BMI) <40 kg/m2.
Exclusion criteria were:
- Incomplete follow-up including non-attendance at post-operative visits and non-completion of both before and after questionnaires (unless documented complete snoring resolution, equivalent to 0/9, in lieu of “after” SSS).
Standard post-operative care included an overnight hospital stay as an inpatient, and 5 days of cefalexin 500 mg qid. Paracetamol was the primary analgesic agent used post-discharge. Patients were followed up at 2 weeks, and again at 3 months and advised to seek care sooner if there were was significant bleeding or any other serious concern.
To quantify the severity of snoring symptoms, the SSS (20) was collected in patients both before and after surgery as a part of regular patient care (3 months post-operatively). The SSS is a short questionnaire which provides information on frequency, nightly duration and loudness of snoring symptoms (20). The primary outcome measure was change in SSS. If there was no record of completion of the SSS questionnaire, but there was documentation in the patient’s clinical notes that snoring had completely ceased, a SSS score of 0 was applied (n=3). The secondary outcome was ESS scores to determine effect on daytime somnolence (21).
Patient demographic data including age at date of surgery, gender and BMI were collated and all polysomnography results had been reported by a qualified sleep physician. Friedman scores collected pre-operatively were also recorded.
For all patients, adverse events following surgery were recorded. For the purposes of this study, post-operative pain lasting less than 1 month and velopalatal insufficiency lasting less than 1 month were considered as common temporary effects of the surgery, not adverse events.
Statistical analysis
Statistical analysis was performed using SPSS 27 (IBM Corp, Armonk, NY, USA 2020). Paired, one tailed t-tests were used to compare pre- and post-operative SSS scores, and unpaired one tailed t-tests were used to compare values where there were incomplete data (BMI, AHI, ESS).
Effect size was calculated using Cohen’s d formula following t-tests meeting the significance threshold.
Results
A total of 97 consecutive patients presenting for snoring treatment underwent modUPPP and RIST between 2008 and 2020 and had an AHI of 15 or less. Seventy-eight (80%) of these patients had completed the SSS prior to surgery and were included in this study, of which 10 were lost to follow-up, resulting in 68 with full data.
The mean age of participants (n=68) was 37.3±11.5 years, comprising 51 males and 17 females. The mean BMI of patients receiving modUPPP surgery was 27.7±4.0 kg/m2, with no statistically significant difference recorded following surgery (P=0.79). The mean AHI prior to intervention was 6.5±4.1. There were no patients with BMI greater than or equal to 40 kg/m2, and 3 patients with BMI greater than or equal to 35. Table 1 provides a summary of pre- and post-operative patient demographics.
Table 1
| Variables | Mean ± SD | Range | P value |
|---|---|---|---|
| Age, years | 37.3±11.5 | 18.6–71.0 | – |
| AHI | 6.5±4.1 | – | – |
| BMI, kg/m2 | 0.79 (NS) | ||
| Pre-surgery (n=59) | 27.7±4.0 | 16.4–39.3 | |
| At follow-up (n=44) | 27.9±4.1 | 16.6–39.5 |
modUPPP, modified uvulopalatopharyngoplasty; SD, standard deviation; AHI, Apnoea-Hypopnea Index; BMI, body mass index; NS, not significant.
The number of included patients in each Combined Friedman Staging System category is provided in Table 2. The number of patients undergoing concurrent airway surgery and type of surgery at other levels is detailed in Table 3. Statistical analyses were repeated excluding patients who also underwent lingual tonsillectomy and prior nasal surgery (e.g., bilateral inferior turbinate reduction), and resulted in no significant change to the outcomes analysed.
Table 2
| Tonsils grade | Tongue grade (No. of patients) | |
|---|---|---|
| 1 or 2a/2b | 3 or 4 | |
| 1 or 2 | 18 | 12 |
| 3 or 4 | 18 | 9 |
| 0 | 3 | 3 |
Friedman stage not recorded clearly in five patients.
Table 3
| Operative Intervention | No. of patients |
|---|---|
| Pre-phase nasal surgery (after polysomnogram) | 2 |
| Previous nasal surgery (before polysomnogram) | 2 |
| Lingual tonsillectomy | 18 |
| Bilateral inferior turbinate reduction (conservative) | 21 |
modUPPP, modified uvulopalatopharyngoplasty.
The SSS reduced from a mean value of 7.0±1.6 to 1.9±2.3 post-operatively [mean difference, −5.1 (95% CI: −5.7 to −4.5)] which was statistically significant (P<0.0001) with a large effect size (Cohen’s d=2.6). There was also a statistically significant reduction in ESS score post-surgery, which decreased from 9.0±4.8 to 4.1±2.8 [mean difference, −4.9 (95% CI: −6.1 to −3.8), P<0.0005], with a large effect size (Cohen’s d=1.2), but patients on average were not sleepy prior to intervention. Despite missing data on post-operative BMI (n=44) there was no significant difference in BMI at 3 months post operatively indicating the surgical outcome may not be attributed to weight loss (Figure 1). Tables 4,5 provide a summary of pre- and post-operative primary and secondary outcome data. Thirteen percent of patients in this sample had complications from the surgery, including globus pharyngeus, velopalatal insufficiency, dysgeusia secondary haemorrhage, and post-operative negative pressure pulmonary oedema (Table 6).
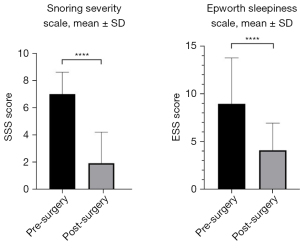
Table 4
| Snoring Severity Scale | Mean ± SD | P value |
|---|---|---|
| Pre-surgery (n=68) | 7.0±1.6 | P<0.0001, effect size 2.6 |
| Post-surgery (n=68) | 1.9±2.3 |
modUPPP, modified uvulopalatopharyngoplasty; SD, standard deviation.
Table 5
| Epworth Sleepiness Scale | Mean ± SD | P value |
|---|---|---|
| Pre-surgery (n=66) | 9.0±4.8 | P<0.0001, effect size 1.2 |
| Post-surgery (n=62) | 4.1±2.8 |
modUPPP, modified uvulopalatopharyngoplasty; SD, standard deviation.
Table 6
| Complication | Lasting between 1–3 months (n) | Lasting up to 6 months (n) | Lasting beyond 6 months (n) |
|---|---|---|---|
| Globus pharyngeus sensation | 1 | 0 | 2 |
| Transient velopalatal insufficiency | 1 | 1 | 0 |
| Dysgeusia | 1 | 0 | 0 |
| Secondary haemorrhage, not requiring readmission or prolonged stay | 2 | 0 | 0 |
| Secondary haemorrhage, requiring readmission or prolonged stay | – | – | – |
| Mild transient negative pressure pulmonary oedema | 1 | – | – |
modUPPP, modified uvulopalatopharyngoplasty.
Discussion
This is the first study to specifically assess the effectiveness of modUPPP and RIST in the management of snoring without significant OSA. The results of this study indicate that there is a major reduction in patient and partner perceived snoring symptoms at follow-up after surgery. Multilevel surgery is effective in decreasing AHI in patients with habitual snoring or OSA of any severity (17,18), with recent studies demonstrating significant reduction in ESS in those with moderate or severe OSA (18). Original uvulopalatopharyngoplasty, first described by Fujita et al. (22), has undergone several modifications to facilitate a reconstructive procedure with reduced morbidity. All patients at our institution undergo pre-operative flexible nasendoscopy in combination with dynamic manoeuvres to determine whether the palate is a contributor to oscillation and noise production. The modUPPP procedure is combined with RIST which stiffens the tongue musculature and improves retro-lingual airway collapse (17,20). It has been previously established that tongue-base snoring produces higher frequency snoring sound and the palate low frequency sound (23), hence both aspects of snoring are potentially treated with a combination of procedures. Furthermore, modUPPP and RIST is an effective treatment for OSA (18) and so this procedure may prevent or impede development of OSA in snorers given it is known that a proportion of primary snorers go on to develop the disease.
Conservative measures such as weight reduction may improve both habitual snoring, and OSA severity, and should be discussed with all patients (24). This study, however, demonstrated no significant change in patient BMI before and after surgery (P=0.79), indicating that the observed improvement in SSS and ESS was due to the effect of surgery rather than any concomitant weight loss.
A comparison between AHI before and after surgery was not made, as this was not a target of the surgery, nor of this study. It was expected that change in AHI would be negligible and not clinically significant, hence it was not sought as a clinical outcome.
In this study, AHI >15 was used to exclude patients. The classical definition of mild OSA has been AHI 5 to 15 (19). In our study patients weren’t sleepy on average (ESS =9.0±4.8), nor did they desaturate significantly, components of OSA that have been linked to medical risk (5,25).
Complications occurred in 9 (13%) patients. Minor complications such as globus pharyngeus (4%), velopalatal insufficiency (3%), dysgeusia (1.5%) were all transient (Table 6). The 2 (3%) secondary bleeds were minor and did not require readmission and the one case of post-operative negative pressure pulmonary oedema (1.5%) was mild and transient, not requiring non-invasive ventilation. No prolonged admissions or readmissions occurred. The complication rate is comparable to that described by MacKay et al. (18) where 12.5% of patients experienced minor surgical or anaesthetic side effects with no post-operative bleeding seen in a higher risk patient cohort (moderate to severe OSA). The literature reports a secondary haemorrhage rate of 4.8% in adult tonsillectomy, with 2.2% requiring some form of intervention (26). This study reports a low rate of bleeding with zero patients requiring return to theatre.
Limitations of this study include, firstly, short-term follow-up. While these results indicate the effectiveness of multilevel surgery in decreasing snoring within this timeframe, studies evaluating maintenance of symptom resolution for longer time periods are needed.
Secondly the use of SSS to determine treatment effect is limited to partner reporting. Further studies should aim to quantify snoring volume, length and frequency by recording methods (27) to improve objectivity of outcome measure, despite the lack of universally accepted acoustic measures.
Conclusions
The results of this study indicate that modUPPP with RIST is an effective treatment modality with a low complication rate. This procedure may be offered to carefully selected patients with snoring (without significant OSA) who have failed or declined device therapy.
Acknowledgments
The abstract of this article has been presented in the 2021 Australasian Sleep Association Abstracts.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://www.theajo.com/article/view/10.21037/ajo-21-47/rc
Data Sharing Statement: Available at https://www.theajo.com/article/view/10.21037/ajo-21-47/dss
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://www.theajo.com/article/view/10.21037/ajo-21-47/coif). SGM serves as an unpaid editorial board member of Australian Journal of Otolaryngology. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the human research ethics committee attached to our local health district [ISLHD Low & Negligible Risk (LNR) Research Review Committee]. LNR reference number: ISLHD/LNR/2021-094, Approval date: 12/02/2021. All patients provided written informed consent prior to surgical intervention. Informed consent was not required for participation in this study, as advised by the ethics committee, given this was a retrospective study based on existing data.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Yunus FM, Khan S, Mitra DK, et al. Relationship of sleep pattern and snoring with chronic disease: findings from a nationwide population-based survey. Sleep Health 2018;4:40-8. [Crossref] [PubMed]
- Dalmasso F, Prota R. Snoring: analysis, measurement, clinical implications and applications. Eur Respir J 1996;9:146-59. [Crossref] [PubMed]
- Marshall NS, Wong KK, Cullen SR, et al. Sleep apnea and 20-year follow-up for all-cause mortality, stroke, and cancer incidence and mortality in the Busselton Health Study cohort. J Clin Sleep Med 2014;10:355-62. [Crossref] [PubMed]
- Landry SA, Banks S, Cistulli PA, et al. A consensus opinion amongst stakeholders as to benefits of obstructive sleep apnoea treatment for cardiovascular health. Respirology 2019;24:376-81. [Crossref] [PubMed]
- Epstein LJ, Kristo D, Strollo PJ Jr, et al. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med 2009;5:263-76. [Crossref] [PubMed]
- Deeb R, Smeds MR, Bath J, et al. Snoring and carotid artery disease: A new risk factor emerges. Laryngoscope 2019;129:265-8. [Crossref] [PubMed]
- Li J, McEvoy RD, Zheng D, et al. Self-reported Snoring Patterns Predict Stroke Events in High-Risk Patients With OSA: Post Hoc Analyses of the SAVE Study. Chest 2020;158:2146-54. [Crossref] [PubMed]
- Beninati W, Harris CD, Herold DL, et al. The effect of snoring and obstructive sleep apnea on the sleep quality of bed partners. Mayo Clin Proc 1999;74:955-8. [Crossref] [PubMed]
- Blumen M, Quera Salva MA, d'Ortho MP, et al. Effect of sleeping alone on sleep quality in female bed partners of snorers. Eur Respir J 2009;34:1127-31. [Crossref] [PubMed]
- Berry RB, Kryger MH, Massie CA. A novel nasal expiratory positive airway pressure (EPAP) device for the treatment of obstructive sleep apnea: a randomized controlled trial. Sleep 2011;34:479-85. [Crossref] [PubMed]
- Berry RB, Block AJ. Positive nasal airway pressure eliminates snoring as well as obstructive sleep apnea. Chest 1984;85:15-20. [Crossref] [PubMed]
- Shapiro GK, Shapiro CM. Factors that influence CPAP adherence: an overview. Sleep Breath 2010;14:323-35. [Crossref] [PubMed]
- Bäck LJ, Hytönen ML, Roine RP, et al. Radiofrequency ablation treatment of soft palate for patients with snoring: a systematic review of effectiveness and adverse effects. Laryngoscope 2009;119:1241-50. [Crossref] [PubMed]
- Brietzke SE, Mair EA. Injection snoreplasty: how to treat snoring without all the pain and expense. Otolaryngol Head Neck Surg 2001;124:503-10. [Crossref] [PubMed]
- Choi JH, Kim SN, Cho JH. Efficacy of the Pillar implant in the treatment of snoring and mild-to-moderate obstructive sleep apnea: a meta-analysis. Laryngoscope 2013;123:269-76. [Crossref] [PubMed]
- Stuck BA, Sauter A, Hörmann K, et al. Radiofrequency surgery of the soft palate in the treatment of snoring. A placebo-controlled trial. Sleep 2005;28:847-50. [Crossref] [PubMed]
- MacKay SG, Carney AS, Woods C, et al. Modified uvulopalatopharyngoplasty and coblation channeling of the tongue for obstructive sleep apnea: a multi-centre Australian trial. J Clin Sleep Med 2013;9:117-24. [Crossref] [PubMed]
- MacKay S, Carney AS, Catcheside PG, et al. Effect of Multilevel Upper Airway Surgery vs Medical Management on the Apnea-Hypopnea Index and Patient-Reported Daytime Sleepiness Among Patients With Moderate or Severe Obstructive Sleep Apnea: The SAMS Randomized Clinical Trial. JAMA 2020;324:1168-79. [Crossref] [PubMed]
- McNicholas WT, Bonsignore MR, Lévy P, et al. Mild obstructive sleep apnoea: clinical relevance and approaches to management. Lancet Respir Med 2016;4:826-34. [Crossref] [PubMed]
- Lim PV, Curry AR. A new method for evaluating and reporting the severity of snoring. J Laryngol Otol 1999;113:336-40. [Crossref] [PubMed]
- Johns MW. Daytime sleepiness, snoring, and obstructive sleep apnea. The Epworth Sleepiness Scale. Chest 1993;103:30-6. [Crossref] [PubMed]
- Fujita S, Conway W, Zorick F, et al. Surgical correction of anatomic azbnormalities in obstructive sleep apnea syndrome: uvulopalatopharyngoplasty. Otolaryngol Head Neck Surg 1981;89:923-34. [Crossref] [PubMed]
- Pevernagie D, Aarts RM, De Meyer M. The acoustics of snoring. Sleep Med Rev 2010;14:131-44. [Crossref] [PubMed]
- Braver HM, Block AJ, Perri MG. Treatment for snoring. Combined weight loss, sleeping on side, and nasal spray. Chest 1995;107:1283-8. [Crossref] [PubMed]
- Marshall NS, Wong KK, Liu PY, et al. Sleep apnea as an independent risk factor for all-cause mortality: the Busselton Health Study. Sleep 2008;31:1079-85. [PubMed]
- Bhattacharyya N, Kepnes LJ. Revisits and postoperative hemorrhage after adult tonsillectomy. Laryngoscope 2014;124:1554-6. [Crossref] [PubMed]
- Arnardottir ES, Isleifsson B, Agustsson JS, et al. How to measure snoring? A comparison of the microphone, cannula and piezoelectric sensor. J Sleep Res 2016;25:158-68. [Crossref] [PubMed]
Cite this article as: Lindsay BM, Sarkis LM, Sideris AW, Lam ME, Jones A, MacKay SG. Modified uvulopalatopharyngoplasty and radiofrequency-in-saline tongue for the management of snoring. Aust J Otolaryngol 2022;5:25.


